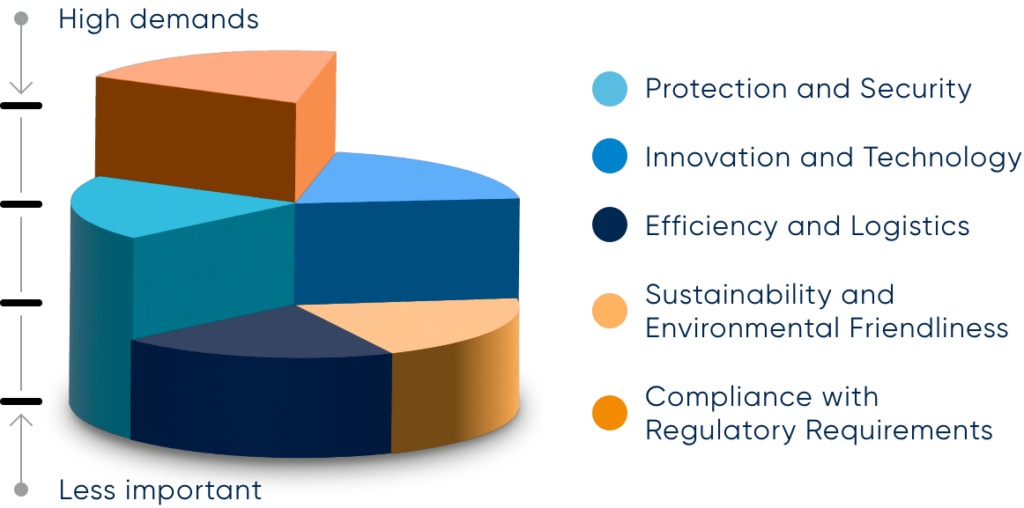
The safe and efficient reprocessing and storage of medical devices requires various aspects to be taken into account. The requirements in the categories ‘Protection and safety’, ‘Innovation and technology’, ‘Efficiency and logistics’, ‘Compliance with regulatory requirements’ and ‘Sustainability and environmental friendliness’ are explained below:








The correct storage and packaging of medical devices requires a careful balance between safety, efficiency and sustainability. Through innovative technologies and compliance with legal requirements, we can ensure the quality of packaging solutions for these high demands.
Services
Industries
Company
Insights

DELTA Packaging Services GmbH
Siemensstraße 8
D-50259 Pulheim
Phone: +49 (0)2238 9491-0
Fax: +49 (0)2238 9491-400
delta.pulheim@dssmith.com
© DELTA Packaging Services GmbH